How Is Nuclear Stability Related to the Neutron Proton Ratio
Elements having atomic numbers greater than 70 are never stable. Nuclear stability makes certain isotopes radioactive.
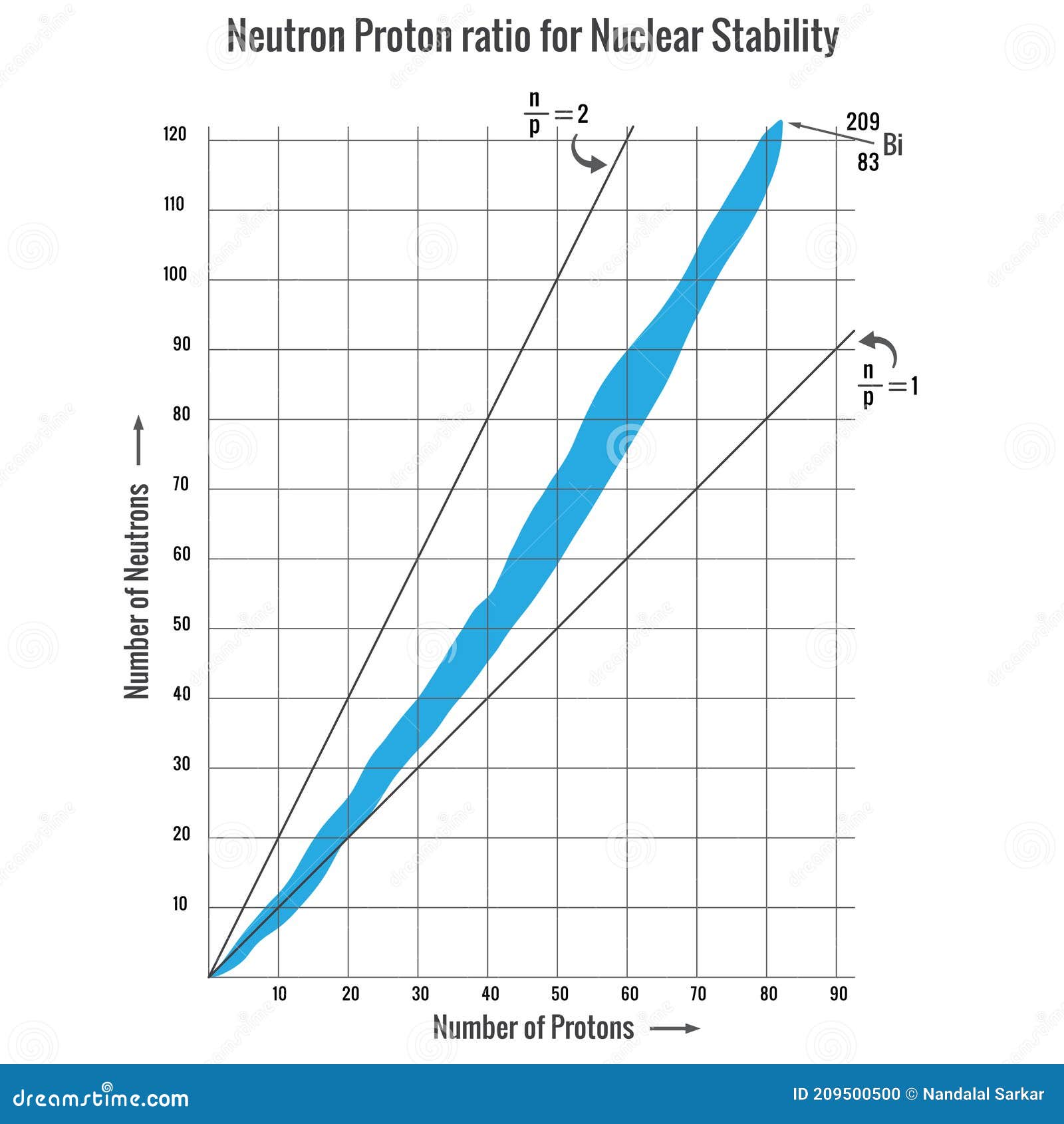
Neutron Proton Ratio For Nuclear Stability Nuclear Reaction Mode Stock Vector Illustration Of Reaction Reactor 209500500
It appears that neutron to proton np ratio is the dominant factor in nuclear stability.
. To identify the stability of an isotope it is needed to find the ratio of neutrons to protons. Nuclear stability depends on. Single neutrons are unstable but in a nucleus they are stable and the more nucleons you have the greater the strong force.
To determine the stability of an isotope you can use the ratio neutronproton NZ. As a result as the number of protons increases an increasing ratio of neutrons to protons is needed to form a stable nucleus. As the nucleus gets bigger the electrostatic repulsions between the protons gets weaker.
Vii Neutron-proton ratio and nuclear stability or causes of radioactivity. X 3 X 2 2 3 H e T are quite exception. One of the simplest ways of predicting the nuclear.
As the number o protons in a nucleus increases the repulsive force becomes freater than the nuclear force. Elements having atomic number less than 20 mostly have proton and neutron ratio 11. Then how do we predict the nuclear stability.
Isotopes that are radioactive because they have unstable nuclei. The np ratio steadily increases as the atomic number increases past element 20 calcium to about element 84 polonium. Neutron to proton ratio The ratio of neutrons to protons np is a successful way in predicting nuclear stability.
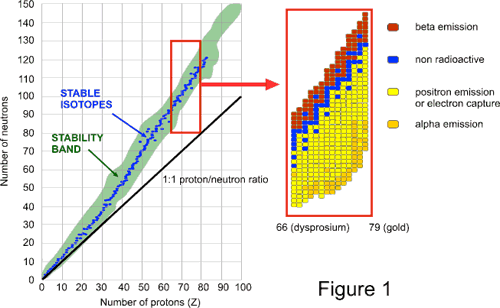
An isotope is called unstable if it has a ratio of protons to neutrons that is not within the band of stability. Also to help understand this concept there is a chart of the nuclides known as. So why cant you have stable isotopes say uranium where excess neutrons increase the binding energy per nucleon ratio to.
They have a neutronproton ratio between 11 and 15. Nuclear Stability is a concept that helps to identify the stability of an isotope. Unstable nuclei attain stability through disintegration.
This ratio is close to 1 for atoms of elements with low atomic number and increases as the atomic number increases. The nuclear strong force is about 100. A certain number of neutrons is needed to offset this repulsive force.
The most stable nuclei have a neutron proton ratio of. Only 1 H and 3 He have neutrons to protons. For nuclei with more than 40.
The nuclear stability is found to be related to the neutronproton np ratio. Im looking for a good description high-school to 1st year level why the ratio of protons to neutrons matters for stability. Stability of an isotope can be determined by calculating the ratio of neutrons to protons present in a nucleus NZ.
The neutronproton ratio NZ ratio or nuclear ratio of an atomic nucleus is the ratio of its number of neutrons to its number of protons. Very few nuclides exist with NZ neutron proton ratio less than 1 NZ. Among stable nuclei and naturally occurring nuclei this ratio generally increases with increasing atomic number.
For stable nuclei np ratio lies close to unity for elements with low atmoic numbers 20 or less but it is more than 1 for nuclei having higher atomic numbers. Nuclei too aside from this ratio See Valley of stability are unstable undergoing beta decay electron or positron emission or electron capture toward the stability valley. About Press Copyright Contact us Creators Advertise Developers Terms Privacy Policy Safety How YouTube works Test new features Press Copyright Contact us Creators.
Nuclei having np ratio either very high or low undergo nuclear transformation. Most of the stable nuclei have neutrons to protons ratio more than 1. Its ratio of neutrons and protons.
Neutrons stabilize the nucleus because they attract each other and protons which helps offset the electrical repulsion between protons. If for different elements the number of neutrons is plotted against the number of protons it is found that the elements with stable nuclei non-radioactive elements lie within a region belt known as zone or belt of stability. If there are too many or too few neutrons for a given number of protons the resulting nucleus.
Unstable nuclei attain stability through disintegration. The number of neutrons increases as the atomic number increases. Radiowaves and visible light are forms of this.
Isobarsthe same nucleon count is generally around 10. Extremely protonneutron rich nuclides may emit a respective nucleon. This ratio is close to 1 for atoms of elements with low atomic numbers of less than about 20 protons.
The nuclear stability is related to neutron proton ratio np. Based on the available stable isotopes. Overview of Nuclear Stability.
An unstable isotope undergoes transmutation alpha decay or beta decay. The stable nuclei are in the pink band known as the belt of stability. For light nuclei till about 40 nucleons the neutronproton ratio of stable isobars the same nucleon count is generally around 10.
For stable nuclei np ratio lies close to unity for elements with low atomic numbers 20 or less but it is more than 1 for nuclei having higher atomic numbers. Nuclei too aside from this ratio See Valley of stability are unstable undergoing beta decay electron or positron emission or electron capture toward the stability valley. The nuclear stability is related to neutron proton ratio np.
Plot of the stable nuclei cluster over a range of neutron proton ratios.

Explain How The Neutron Proton Ratio Is Associated With The Stability Of Two Nuclei Why Is A Certain Ratio Needed Socratic
What Is The Ratio Of Proton To Neutron In Radioactive Material Quora
Comments
Post a Comment